I’ve had an interest in all aspects of science as long as I can remember. Chemistry has always been one of those interests. Organic chemistry began to hold a particular fascination for me due to a pair of related synthetic experiments in my high school organic chemistry class. It’s one thing to discuss theoretical, paper chemistry in a class. It’s another to actually make stuff. That’s when my interest was permanently captured.
The experiments were simple ones, of course. Each began with the same starting material, salicylic acid. Both reactions were esterifications, via different conditions.
The first experiment was an acetylation of the phenolic hydroxyl group of salicylic acid with acetic anhydride to give acetylsalicylic acid (aspirin) as its product. I had known for many years at that time what the chemical name for aspirin was — but I didn’t really have an understanding of what it meant. This changed that.
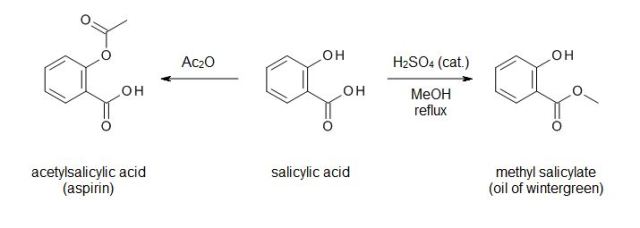 The second was a Fischer esterification (although I don’t recall referring to name reactions in high school) of the same salicylic acid starting material in methanol, to produce methyl salicylate, or oil of wintergreen. The smell of every reaction I would ever again run went downhill after that. (And, of course, you should not deliberately smell the contents of your reaction flask.)
The second was a Fischer esterification (although I don’t recall referring to name reactions in high school) of the same salicylic acid starting material in methanol, to produce methyl salicylate, or oil of wintergreen. The smell of every reaction I would ever again run went downhill after that. (And, of course, you should not deliberately smell the contents of your reaction flask.)
It fascinated me at the time that the same starting material could give products with vastly different properties and uses via a single functional group conversion. To me, this is still one of the cooler parts of organic synthesis — made even cooler when you’re making and characterizing compounds yet unpublished in the scientific literature.
No, I don’t recall what yields I achieved running these reactions. For the sake of argument, we’ll just say it was quantitative. Or a “Swiss yield.” (Bad chemistry joke alert: A Swiss yield is when you leave in the stirbar when weighing your product. I think I first heard that one during a Rick Danheiser seminar. Chemistry humor — not an oxymoron. Believe it!)
Now that I’ve spilled…what aspect of chemistry, or any other science, had you “at hello?”

Dang – your high school had ochem? Even if it was simple ochem, that’s remarkable to me. I can relate to sniffing wonderful, newly synthesized esters for the first time in high school, though. Also, I like “swiss yield” – I’ll pass it on to my friends. 🙂
I’m glad I found your blog!
Yeah, my high school was rare in offering organic chem. It was only a semester’s worth, but I loved it.
Thanks for the feedback — I definitely need to post more often!!